Hey guys,
Kasper sort of got me interested in the idea that perhaps this unique type of display might be repairable. I have a fully functioning Seiko F623 that I'm willing to donate for the test to try and see if it might be possible to get this display working again.
For reference, here's a photo of the watch in question:

The display uses Tungsten Trioxide and Lithium. The Lithium supplies electrons to the Tungsten Trioxide and causes it to turn a vivid blue color. The color becomes more vivid the higher the voltage, up to 1.5v in this case. For reference on how this happens, here's a video that explains how these two chemicals work to make a display like this.:
http://wwwf.imperial.ac.uk/imedia/content/view/559/electrochromic-displays/
****UPDATE****
Had watch sent in to Parsec Enterprises for a cleaning, but after they tried to source parts for the Electrochormic display for about two weeks, they were unsuccessful. Watch was sent back to me in the same condition I sent it. Best advice to others that have this model is to just hold on to them. Perhaps in the future there will be some way to restore the display.
Hopefully some of the digital and/or chemistry guru's will be able to help us come up with a way that the display can be repaired. I suspect that a deposit has formed on the electrodes and that it might be possible to reverse this effect, sort of like how you can electroplate something, but also reverse the effect of electroplating as well. Some of the info I found says that Tungsten Trioxide becomes blue when ions are introduced to it in a current, but that the effect is completely reversible. If you go to far, probably in voltage, the Tungsten will become brown, which is irreversible. So it seems we are in good shape with the display in that regard.
For further info, below is a link on how this display was made with the chemical composition.
http://books.google.com/books?id=p68XfA0MHpoC&pg=PA261&lpg=PA261&dq=Seiko+Electrochromic+watch&source=bl&ots=yp1i-AvrFT&sig=Fjm_TzJ8A9sizvSdiyiNrFFgIVs&hl=en&sa=X&ei=wlPCUOzWL4Wn0AHL44HgDQ&ved=0CDcQ6AEwAg#v=onepage&q=Seiko%20Electrochromic%20watch&f=false
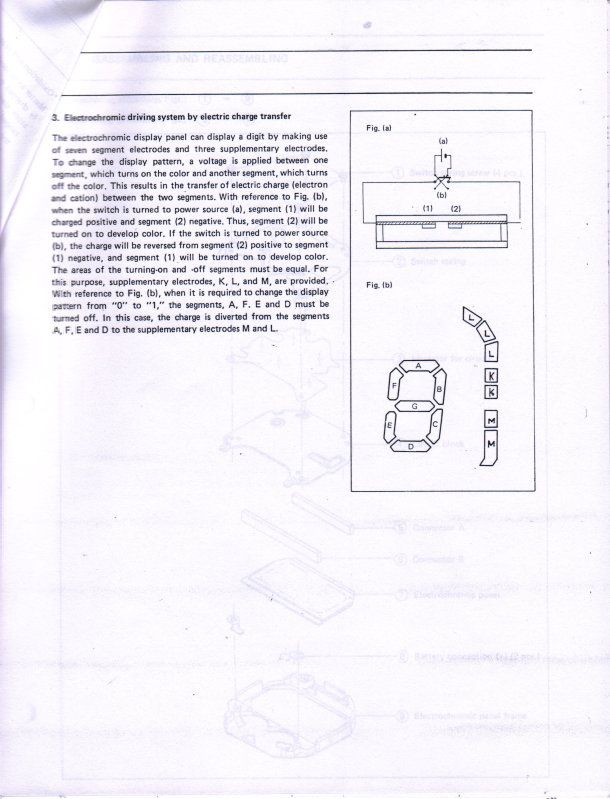
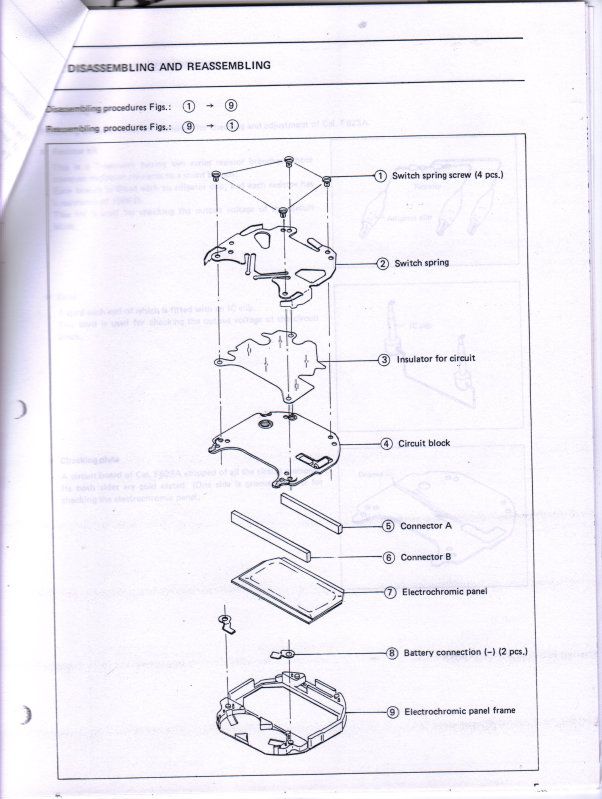
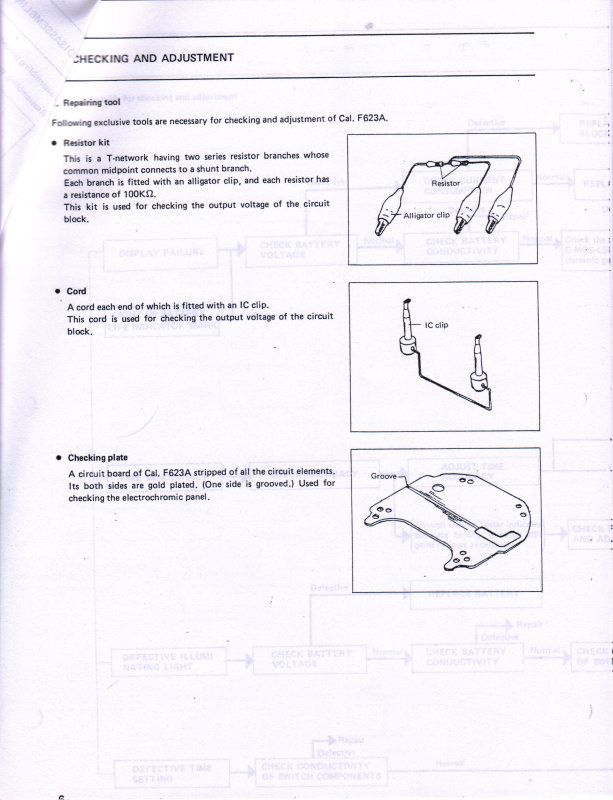
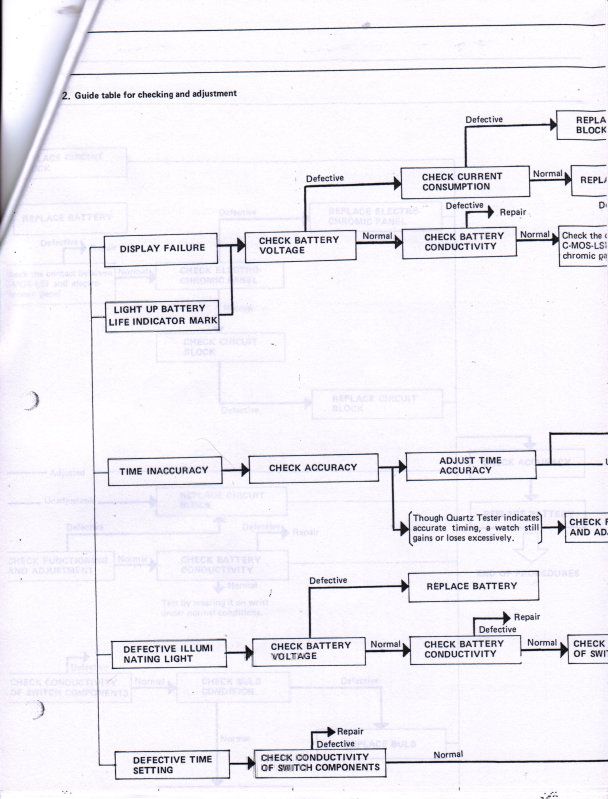
And finally, Seiko was very kind to supply me with their technical repair manual for this Electrochromic display, below is a copy of what they supplied to me to help other's use as a reference for this watch.





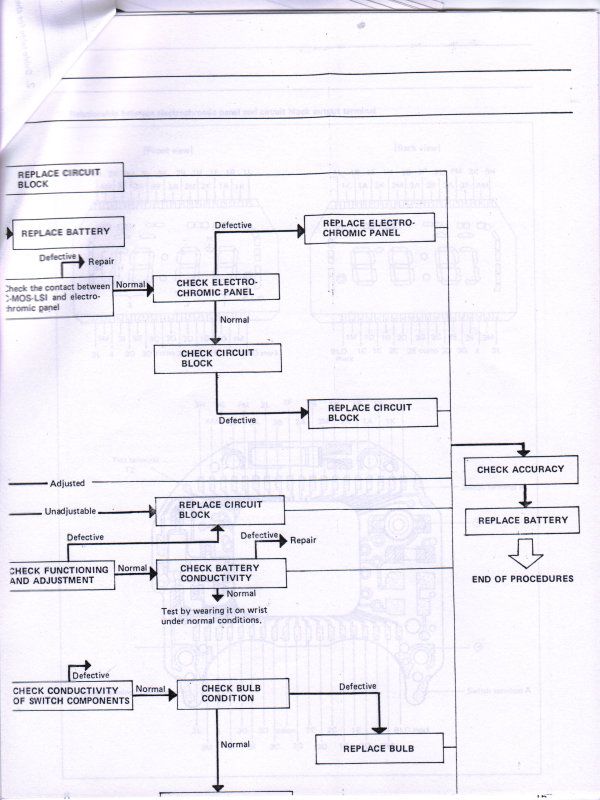
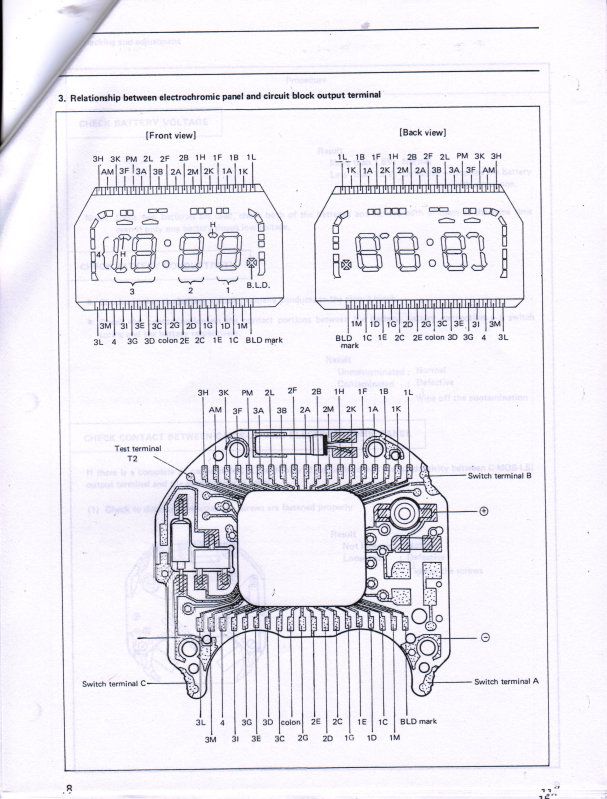

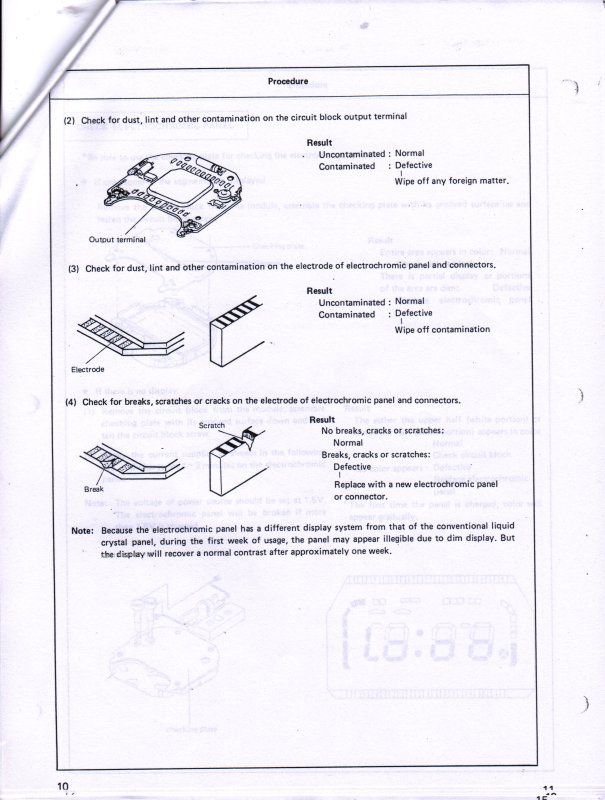




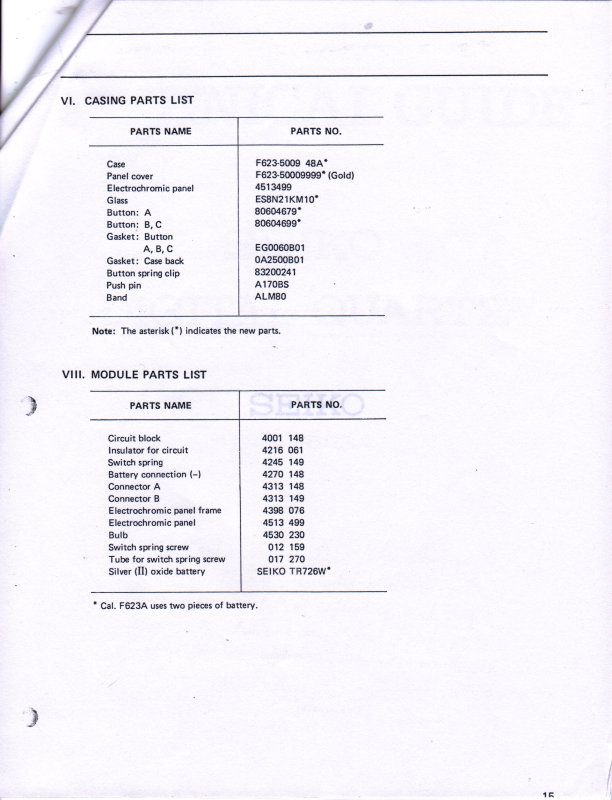
ETA, I found that the book "Repairing Quartz Watches by Henry Fried" Chapter XI is on Electrochromic watches, does anyone have this book and is there any info on what kind of repairs are available?
Additional information provided by Torsten from the book "Repairing Quartz Watches" Thanks Torsten for the info on the rather elusive specifications of the Seiko Electrochromic displays. Links below are direct links to .pdf files on the Seiko F623 testing and specifications.
http://www.4shared.com/office/vrgQ5jxV/hbf1.html
http://www.4shared.com/office/xT_me0ps/hbf2.html
http://www.4shared.com/office/e_iyxQY2/hbf3.html
http://www.4shared.com/office/Qmx549kV/hbf4.html
http://www.4shared.com/office/jKDvC_bi/hbf5.html
http://www.4shared.com/office/QUHtDGry/hbf6.html
http://www.4shared.com/office/ZXJx9GsG/hbf7.html
Kasper sort of got me interested in the idea that perhaps this unique type of display might be repairable. I have a fully functioning Seiko F623 that I'm willing to donate for the test to try and see if it might be possible to get this display working again.
For reference, here's a photo of the watch in question:

The display uses Tungsten Trioxide and Lithium. The Lithium supplies electrons to the Tungsten Trioxide and causes it to turn a vivid blue color. The color becomes more vivid the higher the voltage, up to 1.5v in this case. For reference on how this happens, here's a video that explains how these two chemicals work to make a display like this.:
http://wwwf.imperial.ac.uk/imedia/content/view/559/electrochromic-displays/
****UPDATE****
Had watch sent in to Parsec Enterprises for a cleaning, but after they tried to source parts for the Electrochormic display for about two weeks, they were unsuccessful. Watch was sent back to me in the same condition I sent it. Best advice to others that have this model is to just hold on to them. Perhaps in the future there will be some way to restore the display.
Hopefully some of the digital and/or chemistry guru's will be able to help us come up with a way that the display can be repaired. I suspect that a deposit has formed on the electrodes and that it might be possible to reverse this effect, sort of like how you can electroplate something, but also reverse the effect of electroplating as well. Some of the info I found says that Tungsten Trioxide becomes blue when ions are introduced to it in a current, but that the effect is completely reversible. If you go to far, probably in voltage, the Tungsten will become brown, which is irreversible. So it seems we are in good shape with the display in that regard.
For further info, below is a link on how this display was made with the chemical composition.
http://books.google.com/books?id=p68XfA0MHpoC&pg=PA261&lpg=PA261&dq=Seiko+Electrochromic+watch&source=bl&ots=yp1i-AvrFT&sig=Fjm_TzJ8A9sizvSdiyiNrFFgIVs&hl=en&sa=X&ei=wlPCUOzWL4Wn0AHL44HgDQ&ved=0CDcQ6AEwAg#v=onepage&q=Seiko%20Electrochromic%20watch&f=false
And finally, Seiko was very kind to supply me with their technical repair manual for this Electrochromic display, below is a copy of what they supplied to me to help other's use as a reference for this watch.


















ETA, I found that the book "Repairing Quartz Watches by Henry Fried" Chapter XI is on Electrochromic watches, does anyone have this book and is there any info on what kind of repairs are available?
Additional information provided by Torsten from the book "Repairing Quartz Watches" Thanks Torsten for the info on the rather elusive specifications of the Seiko Electrochromic displays. Links below are direct links to .pdf files on the Seiko F623 testing and specifications.
http://www.4shared.com/office/vrgQ5jxV/hbf1.html
http://www.4shared.com/office/xT_me0ps/hbf2.html
http://www.4shared.com/office/e_iyxQY2/hbf3.html
http://www.4shared.com/office/Qmx549kV/hbf4.html
http://www.4shared.com/office/jKDvC_bi/hbf5.html
http://www.4shared.com/office/QUHtDGry/hbf6.html
http://www.4shared.com/office/ZXJx9GsG/hbf7.html
